Some genetic polymorphisms (present in less than 30% of patients) and environmental factors, such as tobacco exposure, have been identified to increase the susceptibility for developing inflammatory bowel disease (IBD), but it is suspected that there may be other still unknown environmental or epidemiological factors. In this sense, some studies suggested an increased incidence of IBD in individuals undergoing bariatric surgery (BS) for morbid obesity (MO). We aimed to assess whether BS or MO are associated with an increased risk of developing IBD.
MethodsAll individuals resident in Catalonia (7.7 million inhabitants in 2021) with a diagnosis of obesity or MO within the period 2005-2020 were identified from the Catalan Public Health System Database. Children under the age of 18 and those diagnosed with IBD prior to the diagnosis of obesity or MO were excluded. Individuals BS and those with a new diagnosis of IBD were identified, and the likelihood of developing IBD was analyzed by Kaplan-Meier survival analysis. A Cox regression multivariable analysis was performed to assess independent risk factors for the development of IBD, Crohn’s disease (CD) and ulcerative colitis (UC).
ResultsThree cohorts were identified: 94,473 individuals with MO; 1,009,256 with obesity; and 14,698 who underwent BS during the study period. A total of 4,277 new diagnoses of IBD were identified, of which 78 among individuals who underwent BS prior to IBD diagnosis (0.84 cases per 1000 person-years), 409 among individuals with OM but without BS (0.90 cases per 1000 person-years), and 3,790 in obese individuals (0.60 cases per 1000 person-years). The likelihood of developing IBD was significantly higher in patients with MO as compared with obese patients (HR 1.46; 95%CI 1.32-1.62). These differences were maintained when the likelihood of developing CD or UC were assessed separately. In the multivariable logistic regression analysis, MO (HR 1,68; CI95% 1.41-1.99), female gender (HR 1.17; 95%CI 1.05-1.31) and active smoking (HR 1.62; 95%CI 1.43-1.84) were associated with an increased risk of CD. In UC, MO (HR 1,36; 95%CI 1.19-1.55) and BS (HR 2.62; 95%CI 1.34-2.11) were independent risk factors, whereas female gender (HR 0.86; 95%CI 0.79-0.93) was an independent protective factor.
ConclusionMO is an independent risk factor for the development of IBD, for both CD and UC, whereas BS seems to increase the risk only for UC.
Although respiratory failure is the hallmark of severe disease, it is increasingly clear that Coronavirus Disease 2019 (COVID-19) is a multi-system disorder. The presence of gastrointestinal (Gl) involvement by Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) has been suggested by epidemiological, clinical, non-human primate, in-vitro (enteroid) and ex-vivo (human biopsy) studies. Having recently documented persistence of SAR-CoV-2 within the intestinal epithelium 7 months after infection, here we aimed to study mucosal immune cell abnormalities in individuals with prior history of COVID-19.
MethodsIndividuals with previous COVID-19 diagnosis (by either RT–PCR or seroconversion) and controls (without RT-PCR or serological evidence of prior COVID-19 infection) undergoing endoscopic evaluation were recruited into the study (Table 1,2). Colonic and small intestinal (duodenal and ileal) biopsies were analyzed by multiparameter flow cytometry for mucosal immune cell populations including myeloid cells (classical and non-classical monocytes, dendritic cell subsets), T cells (subsets and activation state), B cells (including plasma cells). Persistence of viral antigens was determined by immunofluorescence microscopy (n=30) using a previously published anti-nucleocapsid (NP) antibody.
ResultsThirty subjects with a previous history of COVID-19 (post-COVID), median of 4 months from diagnosis (range 1-10 months), were recruited and compared with 40 normal volunteer (NV) controls. Relative to controls, post-COVID subjects displayed higher frequencies of classical (CD14+) monocytes in both, the colon and the small bowel, while significantly higher frequencies of conventional dendritic cells (cDC) 1 (lin-HLA-DRhiCD14-CD11c+CD141+) and cDC2 (lin-HLA-DRhiCD14--CD11c+CD1c+) were noted in the colon only. Among T cell subsets, CD8+ tissue resident memory T cells (CD8+CD69+CD103+) were significantly increased in colon of post-COVID subjects compared to NV. Among B cell subsets, plasma cells (CD3-CD27+CD38hi) trended higher (p=0.06), while mucosal B cells (CD3-CD19+) were significantly lower in the terminal ileum of post-COVID subjects compared to NV. Finally, with IF, we detected SARS-CoV-2 NP in 10 out of 30 (33%) of post-COVID subjects (Figure 1). There were no significant correlations of these cell populations with either time after the infection or IF positivity.



Innate and adaptive immune cell abnormalities persist in the intestinal mucosa of post-COVID subjects for up to 10 months and may reflect viral persistence or immune cell dysregulation in the intestines. These findings have major implications for understanding the pathogenesis of long term sequela of COVID-19, including long-haul COVID.
Although respiratory failure is the hallmark of severe disease, it is increasingly clear that Coronavirus Disease 2019 (COVID-19) is a multi-system disorder. The presence of gastrointestinal (Gl) involvement by Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) has been suggested by epidemiological, clinical, non-human primate, in-vitro (enteroid) and ex-vivo (human biopsy) studies. Having recently documented persistence of SAR-CoV-2 within the intestinal epithelium 7 months after infection, here we aimed to study mucosal immune cell abnormalities in individuals with prior history of COVID-19.
MethodsIndividuals with previous COVID-19 diagnosis (by either RT–PCR or seroconversion) and controls (without RT-PCR or serological evidence of prior COVID-19 infection) undergoing endoscopic evaluation were recruited into the study (Table 1,2). Colonic and small intestinal (duodenal and ileal) biopsies were analyzed by multiparameter flow cytometry for mucosal immune cell populations including myeloid cells (classical and non-classical monocytes, dendritic cell subsets), T cells (subsets and activation state), B cells (including plasma cells). Persistence of viral antigens was determined by immunofluorescence microscopy (n=30) using a previously published anti-nucleocapsid (NP) antibody.
ResultsThirty subjects with a previous history of COVID-19 (post-COVID), median of 4 months from diagnosis (range 1-10 months), were recruited and compared with 40 normal volunteer (NV) controls. Relative to controls, post-COVID subjects displayed higher frequencies of classical (CD14+) monocytes in both, the colon and the small bowel, while significantly higher frequencies of conventional dendritic cells (cDC) 1 (lin-HLA-DRhiCD14-CD11c+CD141+) and cDC2 (lin-HLA-DRhiCD14--CD11c+CD1c+) were noted in the colon only. Among T cell subsets, CD8+ tissue resident memory T cells (CD8+CD69+CD103+) were significantly increased in colon of post-COVID subjects compared to NV. Among B cell subsets, plasma cells (CD3-CD27+CD38hi) trended higher (p=0.06), while mucosal B cells (CD3-CD19+) were significantly lower in the terminal ileum of post-COVID subjects compared to NV. Finally, with IF, we detected SARS-CoV-2 NP in 10 out of 30 (33%) of post-COVID subjects (Figure 1). There were no significant correlations of these cell populations with either time after the infection or IF positivity.



Innate and adaptive immune cell abnormalities persist in the intestinal mucosa of post-COVID subjects for up to 10 months and may reflect viral persistence or immune cell dysregulation in the intestines. These findings have major implications for understanding the pathogenesis of long term sequela of COVID-19, including long-haul COVID.
Anal ulcerations are frequently observed in Crohn's disease (CD). Their natural history remains poorly known, especially in pediatric-onset CD. The aims of this study were: to determine in a population-based study the risk of anal ulcerations in pediatric onset CD; to identify risk factors for anal ulcerations; to evaluate the risk of progression towards suppurative lesions; to evaluate the risk factors of progression towards suppurative lesions.
MethodsAll patients with a diagnosis of CD before the age of 17 years between 1988 and 2011 within the population-based registry EPIMAD were followed retrospectively until 2013. A specific collection of additional data was performed in patients with anal ulcerations at diagnosis or during follow-up. The variables collected included: proctological examination, diagnostic management (perineal MRI, endoscopic ultrasound, examination under general anaesthesia) and treatment (medical or surgical). Multivariate Cox models were used to identify factors associated with anal ulcerations and factors of progression towards suppurative lesions. An adjusted time-dependent Cox model was used to evaluate the risk of progression of anal ulcerations towards suppurative lesions.
Results1005 patients were included (females, 450 (44.8%); median age at diagnosis 14.4 years (IQR, 12.0-16.1)). 257 (25.6%) had anal ulceration at diagnosis. Cumulative incidence of anal ulceration at 5 and 10 years from diagnosis was 38.4% (CI95%, 35.2-41.4) and 44.0% (CI95%, 40.5-47.2). 
The presence of extra-intestinal manifestations (HR 1.46, CI95% 1.19-1.80, p=0.0003) and upper digestive location (HR 1.51, CI95% 1.23-1.86, p<0.0001) at diagnosis were associated with the occurrence of anal ulceration. Conversely, ileal location at diagnosis was associated with a lower risk of anal ulceration (L2 vs L1 HR 1.51, CI95% 1.11-2.06, p=0.0087; L3 vs L1 HR 1.42, CI95% 1.08-1.85, p=0.0116). Among the 352 patients with at least one episode of anal ulceration, 82 (23.3%) developed perianal suppuration after a median follow-up of 5.7 years (IQR, 2.8-10.6). The risk of perianal suppuration was doubled in patients with anal ulceration compared to those who did not have any ulceration (HR 2.0, CI95% 1.45-2.74, p<0.0001). 
In patients with anal ulceration, the diagnostic period (before or since the “biologic era”), exposure to immunosuppressants and/or anti-TNF did not influence the risk of perianal suppuration.
Anal ulceration is frequent in pediatric-onset CD, with nearly half of patients presenting with at least one episode after 10 years of evolution. Perianal suppurations are twice as frequent in patients with present or past anal ulceration. These results plead for a proactive therapeutic approach in case of anal ulcerations.
Ulcerative colitis and Crohn’s are both chronic inflammatory bowel diseases, associated with an increased risk of colitis-associated cancer secondary to the longstanding and severe intestinal inflammation. To reduce this risk patients are treated with drugs such as immunomodulators and biologicals. Studies however have shown that these therapies may be associated with an increased risk of extra-intestinal cancers as e.g. haematological malignancies, skin cancer, cervix cancer. Different types of drugs are associated with different types of cancer, e.g. patients treated with thiopurines are at risk of haematological malignancies, such as lymphomas, and non-melanoma skin cancer, whereas a relationship between melanoma and biologicals has been described. Therefore, the benefits and harms must be considered in treatment decision. To reduce the risks of malignancy it is advised temporarily interrupt treatment or restrict the use of certain drugs to a limited time. To avoid skin cancer sun protection and skin surveillance is recommended.
Many neoplastic complications are described in IBD these could be due to the desease or to the treatment. In this presentation we will focus on a case of multiple neuroendocrine tumors in a patient with Crhon's disease.
Educational objectives:
- to know the main neoplastic complications in IBD
- to understand the possible etiology.
To understand neuromuscular complications in IBD including secondary / medication induced changes and extraintestinal involvement of neuromuscular system.
In the last week of February 2020 hit Stockholm with increasing number of cases starting to appear at hospitals in the region. This prompted a rapid mobilisation of large scale biobanking and immediate immunological analysis at the Karolinska Institutet with the aim to understand COVID-19 disease pathogenesis. Fairly soon the Karolinska COVID-19 Immune Atlas was launched as an online resource providing scientists rapid access to biological data. In this talk, the research response to the pandemic will be discussed.
Educational objective:
To undertand the role human translational immunological research have in shedding light on disease mechanisms
1) Description of immune pathways that drive inflammation in IBD
2) Discussion of the pathways targeted by current and future therapies
3) review of clinical evidence supporting use of novel therapies (selective Jaki, Sphingosine modulators, anti p19 therapies)
4) Highlight the data on adverse effects of new therapies
5) Stem cell therapies for perianal fistulae
6) Discuss novel formulations of existing agents (low systemic bioavailable steroids)
1. To understand the role of advanced endoscopic technologies for disease activity and mucosal healing assessment in UC.
2. To review the new Virtual Electronic Chromoendoscopy 'The PICaSSO''score in UC and its correlation with histology
3. To review the new PICaSSO Histological Remission index in UC and its practicality
3. To emphasise the implementation of the PICaSSO endoscopic and histological scores into Artificial Intelligence
In order to guide therapy in Ulcerative Colitis (UC), repeated determination of intestinal inflammatory activity is essential. Endoscopy is the standard procedure to assess inflammation in UC. However innovative methods for non-invasive, uncomplicated and risk free estimation of inflammatory activity are needed as bowel preparation, patients discomfort and risk of procedural complications limit the (frequent) use of colonoscopy. Multispectral optoacoustic tomography (MSOT) is a promising new method to measure inflammation in UC. Using short and harmless impulses of NIR-lasers, it allows for determination of a specific hemoglobin-signal in the bowel-wall and therefore inflammatory activity in affected bowel segments. However, its informative value in UC has not been evaluated so far.
MethodsIn 34 patients with confirmed UC, clinical activity parameters (e.g. clinical Mayo-Subscore, B-mode-sonography, C-reactive protein, white blood count) were collected and MSOT of the sigmoid was performed within 2 weeks before/after endoscopy. For MSOT, a commercially available clinical MSOT-system (Acuity Echo, iThera Medical, Munich) was used with sequential analysis of collected data on an external desktop PC. Finally, clinical data, ultrasound findings (Limberg) and MSOT-parameters (single wavelenghts 760 nm, 800 nm, 900 nm; multispectral signals hb, hbO2, hbT) were correlated with endoscopic findings (Mayo endoscopic Subscore, MES).
ResultsWe found strong and significant correlation between MES and MSOT parameters 800 nm (Spearman r = 0,6599; p < 0,0001) and HbO2 (Spearman r = 0,6695; p < 0,0001), superior to sonographic evaluation of the inflammatory activity in affected bowel segments (Spearman r = 0,4914; p = 0,0023) . Simultaneously these MSOT parameters demonstrated excellent sensitivity and specifity in distinguishing moderately to highly active (MES 2,3) from inactive and mild disease (MES 0,1) (800nm: AUROC 0,9063 (p < 0,0001); sensitivity = 93,75 %, specificity = 88,89 %; HbO2: AUROC 0,9063 (p < 0,0001); sensitivity = 100 %, specificity = 88,89 %).
ConclusionMSOT is a promising approach to non-invasively assess intestinal inflammation in UC and therefore monitor anti-inflammatory therapy in these patients. Further studies are required to validate these findings.
Inflammatory bowel diseases may lead to many non-neoplastic intestinal and extra-intestinal problems.
The main intestinal complications comprise infections, obstruction, perforation fistulas etc, whereas the main extra-intestinal manifestations include skin conditions, liver and biliary diseases, eye complications, systemic infections and other systemic conditions.
Learning objectives:
- which are the main non-neoplastic intestinal complications
- which are the main non-neoplastic extra-intestinal complications
- example and presentation of interesting histological cases
Discussion of non pharmacological strategies to maintain remission
Review of evidence for dietary intervention
Pre and probiotic therapy
Complementary therapies
Modifying the faecal microbiota
Techniques to modify stress
Vagal nerve stimulation
The efficacy of risankizumab (RZB), an interleukin 23 p19 inhibitor, in patients with Crohn’s disease (CD) has been reported. Normalisation of high-sensitivity C-reactive protein (hs-CRP) and faecal calprotectin (FCP) are intermediate treatment targets in CD. Here, we evaluated changes in these objective inflammatory biomarkers and clinical outcomes with RZB treatment.
MethodsIn 2 phase 3, randomised, double-blind studies (ADVANCE, NCT03105128; MOTIVATE, NCT03104413), patients with moderately to severely active CD received 12-week intravenous (IV) RZB induction therapy or placebo (PBO). Patients with clinical response to RZB IV induction were rerandomised in a 52-week maintenance study (FORTIFY, NCT03105102) to receive subcutaneous (SC) RZB or PBO (ie, RZB withdrawal). Induction analyses included patients who received either 600 mg RZB IV or PBO for 12 weeks. Maintenance analyses included patients who received 360 mg RZB SC every 8 weeks or withdrawal (PBO SC) for 52 weeks. Outcomes assessed were normalisation of hs-CRP and FCP concentrations at week 12 of induction and at week 52 of maintenance in patients with elevated biomarkers at baseline (hs-CRP > 5 mg/L and/or FCP > 250 μg/g), clinical biomarker response (defined as enhanced clinical response [≥ 60% average daily stool frequency (SF) decrease and/or ≥ 35% average daily abdominal pain score (APS) decrease] and ≥ 50% reduction in hs-CRP or FCP), and clinical biomarker remission (defined as clinical remission per CD Activity Index or SF/APS criteria and normal hs-CRP or FCP) during maintenance treatment. Nonresponder imputation was used for missing data.
ResultsGreater proportions of patients receiving RZB vs PBO achieved normalisation of hs‑CRP and FCP at the end of the 12-week induction period and the 52-week maintenance period (P < .0001 for all; Table). Among patients with clinical response to RZB IV induction and entered maintenance, rates of clinical biomarker response were maintained through week 52 in patients receiving 360 mg RZB SC and declined over time among patients in the withdrawal (PBO SC) arm (Figure A). Rates of clinical biomarker remission increased over time in patients receiving 360 mg RZB SC. At week 52, clinical remission and normalisation of hs‑CRP or FCP was achieved by 41% of patients receiving RZB vs 28%–29% of patients in the withdrawal (PBO SC) arm (Figure B-C). The safety profile of RZB in CD was previously reported.

Normalisation of objective biomarkers of inflammation in CD was achieved with RZB induction and maintenance therapy. Improvements in both clinical outcomes and biomarker levels were sustained with continuous RZB maintenance therapy and decreased over time in patients discontinuing RZB.
To explore the role and scope of the IBD nurse.
To emphasise the importance of the impact and perspective of IBD in our patients.
To overview the UK IBD standards and how they enhance the provision of quality of care for all patients with IBD.
To provide an overview of the N-ECCO Consensus statements and the different levels in IBD nursing.
Objectives:
•To discuss why should we perform a nutritional assessment in patients with IBD
•To demonstrate how to perform a nutritional assessment
•To emphasize who and when should be screened
•To understand the importance of working together in a multidisciplinary team
1. To provide an overview of some of the nutrition and dietary management options for IBD
2. To highlight the role of the IBD nurse in relation to nutrition and dietary management of IBD
1. To establish a standard terminology for the elaboration of trusted reports in endoscopy, surgery and histopathology
2. To provide i interpretation guidance for dysplasia and cancer, surgical indication and strategies including margins and extent of resection, assessment of disease activity.
3. To standardise the interpretation of biopsies and specimens.
4.To prove useful these recommendations in daily care of IBD patients
Disruption of immune cell trafficking via integrins is a proven and effective mechanism for treating inflammatory bowel disease. When α4β7 integrin is inhibited through pharmacological intervention, immune cells destined for the gut tissue become sequestered in blood circulation and these alterations can be detected through several methods. MORF-057 is a novel, oral, selective, small molecule inhibitor of α4β7 integrin developed for treating IBD. MORF-057 demonstrated favorable tolerability, pharmacokinetic and pharmacodynamic profiles including saturating receptor occupancy and corresponding evidence for proof of biology based on effects on circulating cells during a Phase 1 clinical trial in healthy volunteers (Ray, ECCO 2021). Here we demonstrate an exposure:response relationship of α4β7 related biomarkers examined among MORF-057 treated non-human primates (NHPs) as a means for pre-clinical testing of inhibitors of this pathway.
MethodsSingle-cell RNA sequencing (scRNAseq) was performed on NHP CD45+ blood cells to determine baseline populations potentially impacted through exposure to MORF-057. 40 Naïve cynomolgus monkeys were enrolled over 5 separate studies and dosed orally BID with MORF-057 over 2-7 days using several dose levels to examine biomarker dynamics over a wide range of exposures. Peripheral blood was sampled at various timepoints (n= 125) and assayed using: mass spectroscopy, flow cytometry (FACS), and mRNA quantification to determine MORF-057 exposure levels, on-target receptor occupancy (RO), immune cell subset changes, and CCR9 mRNA levels.
ResultsMORF-057 Ctrough ranged from 3.3-429 ng/ml. In samples tested for RO, MORF-057 achieved >95% saturation of α4β7 even with the lowest Ctrough of 4.5 ng/ml. MORF-057 treatment led to significantly sustained increases in circulating β7high CD4+ T memory cells detectable as early as 24h post-exposure. A maximal effect where β7high cells accounted for approximately 60% of the T memory population was achieved at saturating receptor occupancy (Fig., left). CCR9 mRNA values demonstrated increases upon drug exposure (Fig., right).
In NHP, acute changes in circulating β7high T memory cells was a sensitive biomarker demonstrating a dose-dependent response to MORF-057 exposure. CCR9 mRNA levels also showed similar exposure related changes reflecting its expression on a subset of β7high cell types. Analysis of scRNAseq shows expression of α4β7 on other cell types beyond T memory cells including: NK, NKT, B cells, plasmablasts, monocytes, and eosinophils. Pharmacodynamic changes in NHP are consistent with human Phase 1 data in healthy volunteers.
Limited data are available on the use of Vedolizumab (VDZ) in paediatric Crohn’s Disease (CD) and Ulcerative Colitis (UC). We evaluated the effectiveness and safety of VDZ to induce remission at week 14 in the prospective, multicenter VEDOKIDS study.
We enrolled children (age 0-18 years) with CD or UC commenced on VDZ with a standardized dosing of 177mg/BSA up to 300mg at 0, 2, 6 and q8 weeks thereafter. Non-responders had their dose escalated to q4wks at the discretion of the local physician. Explicit demographic, clinical and safety data were prospectively recorded via REDcap. Clinical remission was defined as steroid- and EEN-free remission (i.e. wPCDAI<12.5 or PUCAI<10) without the need for new medications. Complete remission was defined as clinical remission with normal CRP and ESR. Predictors of response were explored by Logistic regression.
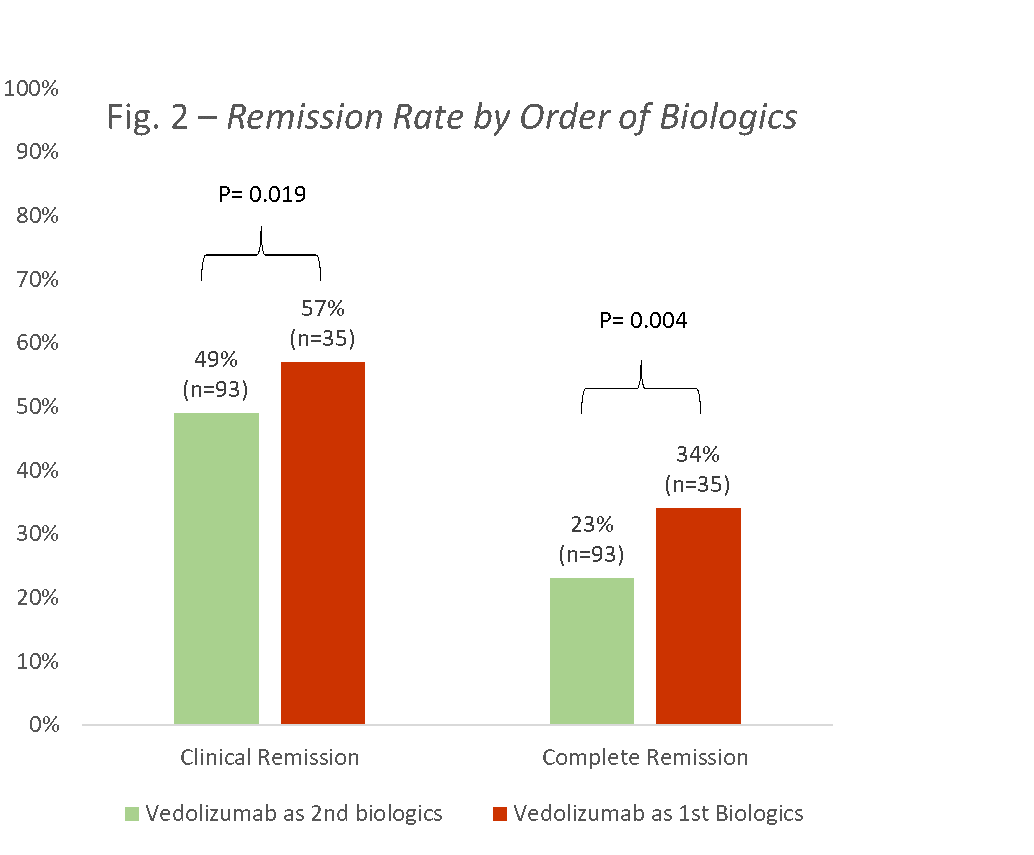
Results128 children were enrolled, 60 (47%) with CD, and 68 (53%) with UC (58 (45%) males, mean age 13.8±3.6, 93 (73%) failed previous anti-TNF, median disease duration 2.3 years (IQR 0.9-4.7)). Using the ITT principle, clinical and complete remission rates for CD at week 14 were 30% and 20%, respectively, and for UC 50% and 38%, respectively (Fig 1). Clinical remission rates of those receiving VDZ as first line biologics versus second line were 57% and 34%, respectively (p=0.019; Fig 2); the corresponding complete remission rates were 49% and 23% (p=0.004).
In the UC group, disease activity at baseline measured by the PUCAI predicted clinical remission at week 14 (OR=0.95, 95%CI 0.93-0.98; median baseline PUCAI 15 (IQR 0-30) in those achieving remission and 45 (20-55) in those who did not; p=0.002). ESR (OR=0.94, 95%CI 0.89-0.98; p=0.009) and a trend towards extensive disease (L3 vs. L1 and L2; OR 0.14, 95%CI 0.18-1.036, p=0.054) predicted clinical remission in CD.
During the 14 weeks, 113 adverse events (AE) were recorded in 58 children: 28 AEs were possibly related to VDZ, all of which were mild-moderate and only 3 (11%) led to discontinuation of VDZ (leukocytoclastic vasculitis, myalgia and dyspnea). There were 18 serious AEs, only one was graded as possibly related to VDZ (headache). There were 18 non-serious cases (19%) of upper respiratory infections (pharyngitis, tonsilitis, parotitis, and otitis media) and one Campylobacter jejuni which was graded as serious.

In this prospective multicenter study, VDZ was safe and effective for inducing remission in a refractory cohort of paediatric IBD, more so in UC. Disease severity and extent at baseline may predict clinical response.


